 
You may change your mind as often as you wish. (in case your answer gets used in class). 
Please type the last five digits of your ID number: Question 13 of 13 What mass in grams of NaN, is required to produce 50.2 L of N, gas (density = 1.25 L according to the balanced chemical reaction: 2 NaN,(s) - 2 Na(s) + 3 N (9) х STARTING AMOUNT ADDINGTON WEWER RESET 28.02 1 N 146 77.7 218 22.99 65.02 502 3 97.1 6.022 x 10 1.This assignment was due by noon on Tuesday, March 11, 2003. Question 12 of 13 How many grams of nitric acid. 
< Question 71 of 13 How many moles of Mno, are produced when 4.30 kg of oxygen gas completely reacts according to the balanced chemical reaction: 2 Mn() 30,(0) - 2 MnO,(6) X STARTINO AMOUNT ACOTACHOR MWEN 0.0896 1 0.001 4.30 32.00 89.6 6.022 x 10 2 3 54.94 102.94 1000 9.22 10° molo, 90, mol Mno као M mol Mn Mno, Question 10 of 13 < How many grams of oxygen gas are produced when 2.43 * 10g of KCIO, are completely reacted according to the following chemical equation: 2 KCIO, (S) - 2 KC(s) + 30,0) х O STARTING AMOUNT ALIOTACION AIMER REY ( ) 2 32.00 1 1.90 104 74.56 6.022 * 10-9 2.97 * 109 5.95 * 10 122.56 198 * 10 9.62 10 2.43 x 10 2 3 mol KCIO GKCI GKCIO, mol KCI gº molo, < Question 9 of 13 Determine the number of molecules of Cr, that form when 1.34 * 10g of oxygen completely reacts according to the following equation: Cr(s) + 3 0,0) - 2 0,0,(s) х STARTING AMOUNT ADD FACTO ANSWER SET 2 152.00 1.34 x 10° 2.52 * 10" 3 6.022 x 10 52.00 4.24 x 10° 32.00 1.68 x 10" 1 4 3.78 x 10 g61,0 molecules Cro g Cr mol mol Cro go mol CrĬhemio € → C. Question 8 of 13 Determine the number of grams of HCl that can react with 0.750 g of Al(OH), according to the following reaction Al(OH),(s) + 3 HCl(aq) - AIC (aq) + 3 Ho(aq) х STARTING AMOUNT ADDINGTON ANIMER ET 1 2.25 0.750 78.00 0.0288 0.351 133.33 36.46 3 18.02 6.022 x 10 9.60 x 10" 1.05 0.117 GAICI, mol HO GAICI GHCI mol HCI mol Al(OH), mol AICI GHO g Al(OH), mol AICI < Question 7 of 13 Determine the number of grams of CH, that are required to completely react to produce 8.70 Mol of Co, according to the following combustion reaction: 2 CH (0) 130,00) - 800,0) 10 H0(0) T STARTINO AMOUNT ADOTACE AIMER SET 58.14 18.02 208.00 0.0374 126 2 8.70 44.01 4.35 6.022 x 10 2.18 13 8 10 mol CH, molco mol H0 SCH, GHO 90, molo gco, Question 6 of 13 How many moles of H,SO, are required to completely react with 7.20 mol of Al according to the balanced chemical reaction: 2 Al(s) + H.S0 (aq) - AI,(SO),(aq) + 3 H (9) х STARTING AMOUNT ADD FACTOR ANIWER REBET N 3.60 26.98 98.08 10.8 6.022 * 10" 21.6 2.40 2.02 3 7.20 342.14 GAI gH,SO mol Al,(50.), Al,(50), mol Al mol H,SO gH, molH Question 5 of 13 mol of B How many moles of Care formed upon complete reaction of 2 according to the generic chemical reaction: X STARTING AMOUNT ADO FACTOR ANSWER 2 0.5 4 1 2 6.022 * 10 mol A molc mol B Calculate the theoretical yield in moles NH from the complete reaction of 15.6 grams in the presence of excess N, gas according to the following balanced chemical equation: No 3 H(o)- 2 NH (0) х STARTING AMOUNT ADORACIOR AIMER REDET ( ) 28.02 1.01 0.612 2 0.918 2.57 87.7 0.306 17.04 14.01 1 7.72 11.6 3 15.6 5.15 10.3 2.02 197 gH g/mol g/mol NH mol NH mol GNH Question 4 of 13 r Consider the Haber-Bosch process for the synthesis of ammonia from its elements. 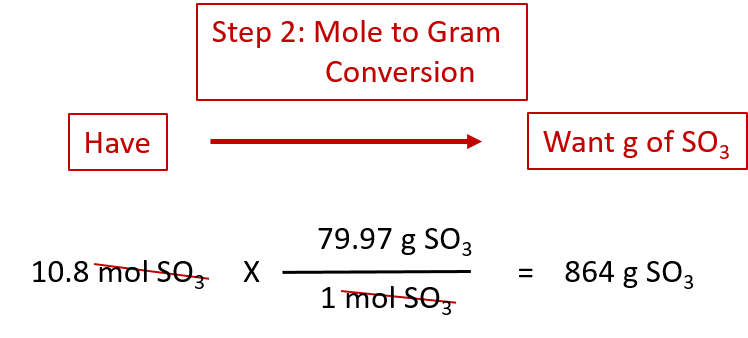
Calculate the theoretical yield in moles NH, from the complete reaction of 59.8 grams in the presence of excess H, gas according to the following balanced chemical equation: N(g) + 3 H (9) - 2 NH (0) х STARTING AMOUNT NO FACTOR NNGIR 2 2 2.13 72.7 14.01 8.54 3 1.01 28.02 59.8 145 4.27 17.04 1 3.52 7.04 g/mol NH, mol N g/mol mol NH, gN NH, Question 3 of 13 Consider the Haber-Bosch process for the synthesis of ammonia from its elements. Question 2 of 13 Calculate the theoretical yield in grams All from the complete reaction of 113 grams, according to the following balanced chemical equation: Al(s) + 3) - 2 All (8) х STARTING AMOUNT ADO FACTOR AJWER RESET ) - 2 113 60.5 0.297 407.68 0.445 253.80 3 121 All g/mol mol Allg/mol All mol! < Question 1 of 13 Calculate the theoretical yield in grams All, from the complete reaction of 18.3 grams Al according to the following balanced chemical equation: 2 A(s) + 3) - 2 All,(s) 407.7 SANI 18.3 GAI 1 277 g All STARTING AMOUNT 26.98 mol All 18.39A 407.7 All 26 98 mol All 277 g Al ADO FACTOR ETE ANGOR PESET it 18.3 407.7 1.47 1 26.98 2 0.678 277 3 138 mol All mol A All, o'mol All GA g/mol A 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
